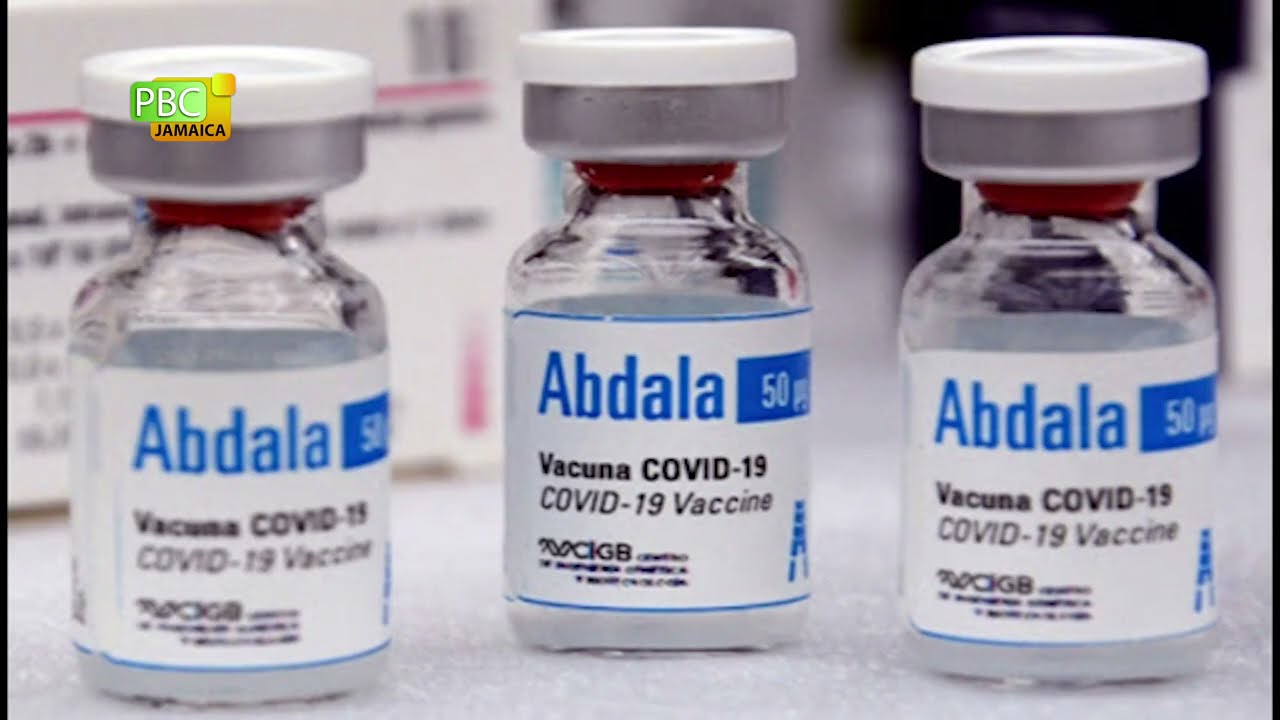
Cuba on Friday approved its home-grown Abdala coronavirus vaccine for emergency use, the first for a Latin American country.
The CECMED health regulator gave the go-ahead after Abdala’s makers last month announced the vaccine candidate was more than 92 per cent effective at preventing COVID-19 after three doses.
Cuba is working on five coronavirus vaccines, and in May started immunizing its population using two of them — Abdala and Soberana 2, then still undergoing clinical trials.
In June, the BioCubaFarma laboratory said Abdala “shows efficacy of 92.28 per cent in its three-dose scheme”.
On Thursday, the Finlay Institute which makes Soberana 2 said it, too, showed efficacy exceeding 91 per cent after three doses.
The World Health Organization has set a 50-per cent efficacy threshold for coronavirus vaccines to offer protection against the virus or disease.
Cuba has been relatively unscathed by the outbreak but has seen a recent sharp increase in cases, with 218,000 cases among its population of 11.2 million, and some 1,450 deaths.

COMMENTS